Establish a unified B2B2C workflow and eliminate data silos across the entire referral-patient-device lifecycle, through formalized templates, automations, and integration readiness.
If this demo appears blurry: increase the Quality to 1080p+ by hovering over the playing video and clicking the gear icon.

Nurture referral sources to identify areas that generate new business and streamline patient access
Establish relationships across inventory, devices, and patient for regulatory, support, and maintenance

Quickly respond to patient or referral support requests and questions coming from multiple channels (webform, email, phone)

Capture the information accurately and completely to start the workflow, including patient and reporter check-ins, internal analysis, and integrate with the QMS system
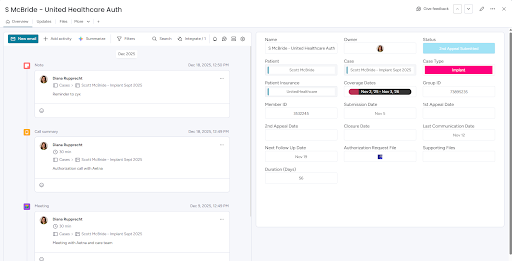
Automated status tracking and support workflows to gather documentation, maximize coverage, and follow-up with insurance during appeals to prove medical necessity
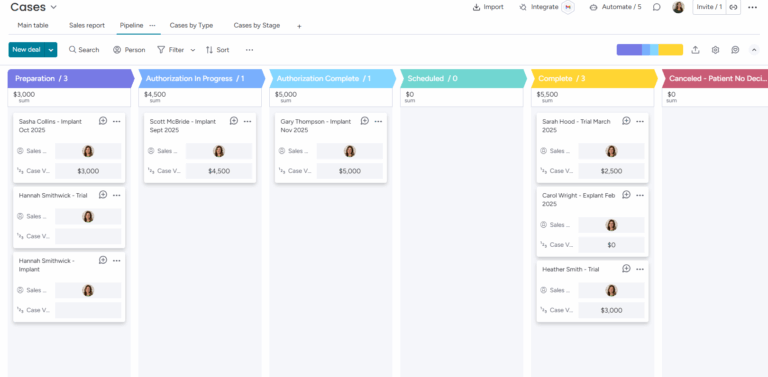
Prospective patients are tracked through initial meetings and qualification steps, and converted to patients once they meet specific criteria.
Patient events such as implantation, explantation, trial, or an order/send for a wearable device.
Functions as a deal record or opportunity, carrying a dollar value for sales forecasting purposes.
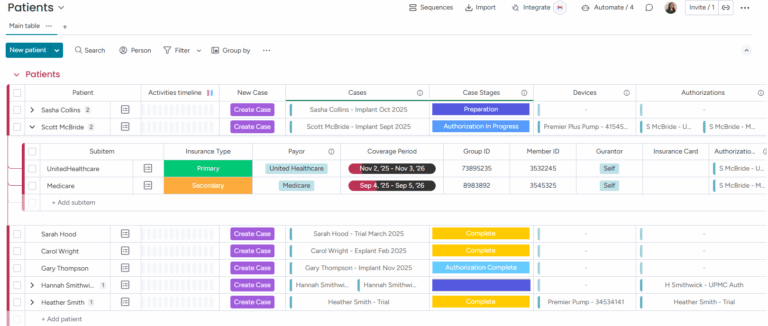
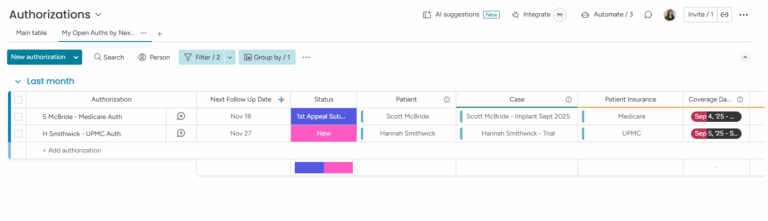
Authorization records are automatically created for each new case and linked to the patient's specific insurance provider.
Critical for compliance tracking and is necessary because devices are expensive and often require multiple submissions and documentation to obtain pre-authorization.
Automation sets follow-up dates after submission to help patient access coordinators manage open authorizations and work through the arduous process.
Specific devices, tracked by serial number, are linked to a case and a patient, along with status updates and milestones.
Devices are tracked for their current location, including shelf stock, trunk stock, and implanted status.
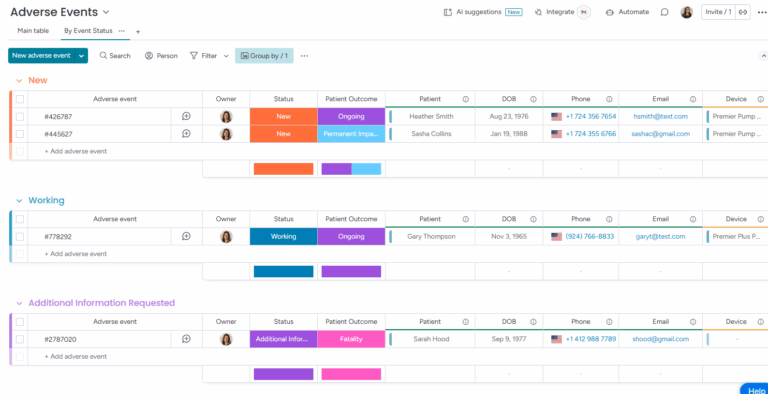
Adverse event records track major compliance issues, typically involving injury or harm caused by a device malfunction.
Complete the form below for more information or a custom demo.





































